PPTX
Module 2 N609 Fall 2025
Module 2: Pharmacogenomics & Precision Medicine
Original source file is included in this package; the embedded viewer and full extracted text are available below.
Slide Text Extract
Improved reading view: slide text is enlarged, spaced out, and placed in expandable study cards. Use Expand all to read continuously, or search the whole textbook from the search box.
Images embedded in the deck are shown after the slide text. Open the original PPTX for exact slide layout.
Slide 1
Pharmacotherapeutics for Advanced Practice:
Module 2
Slide 2
Pharmacogenomics
Slide 3
Learning Objectives
Explain the basic concepts of pharmacogenomics and be familiar with its terminology.
Discuss how genetics affect the activity of drug-metabolizing enzymes.
Consider how pharmacogenomics may play a role in drug therapy selection given a specific case.
Discuss how genetics affect the activity of drug-metabolizing enzymes.
Consider how pharmacogenomics may play a role in drug therapy selection given a specific case.
Slide 4
Pharmacogenomics and Precision Medicine
Pharmacogenetics: refers to single or a few gene variations (called polymorphisms).
Pharmacogenomics: refers more broadly to the genome-wide (or an individual's entire deoxyribonucleic acid [DNA] sequence) effects on drug therapy.
Pharmacogenomics: refers more broadly to the genome-wide (or an individual's entire deoxyribonucleic acid [DNA] sequence) effects on drug therapy.
Slide 5
Personalized Medicine
It includes pharmacogenetics/pharmacogenomics.
It refers to "an emerging practice of medicine that uses an individual's genetic profile to guide decisions made in regard to the prevention, diagnosis, and treatment of disease" (National Human Genome Research Institute, 2020).
This term is evolving to "precision medicine."
Much of the progress made in this field focused on pharmacogenomics.
It refers to "an emerging practice of medicine that uses an individual's genetic profile to guide decisions made in regard to the prevention, diagnosis, and treatment of disease" (National Human Genome Research Institute, 2020).
This term is evolving to "precision medicine."
Much of the progress made in this field focused on pharmacogenomics.
Slide 6
Basic Concepts #1
The human genome is the underpinning to every human's individuality.
With the exception of identical twins, the genome is different for every individual, although there are only small differences among people's DNA that make them unique.
The human genome consists of approximately three billion base pairs, 99.9% of which are the same among all humans with only 0.1% variation among individuals.
With the exception of identical twins, the genome is different for every individual, although there are only small differences among people's DNA that make them unique.
The human genome consists of approximately three billion base pairs, 99.9% of which are the same among all humans with only 0.1% variation among individuals.
Slide 7
Basic Concepts #2
The variations that occur with DNA (polymorphisms), along with environmental and dietary factors, create a patient's individuality, susceptibility to disease, and response to treatments.
Included in this individuality is a person's ability to absorb, distribute, metabolize, and excrete drugs.
Understanding the genetic components affecting these pharmacokinetic processes can help the clinician tailor treatment for a patient.
Included in this individuality is a person's ability to absorb, distribute, metabolize, and excrete drugs.
Understanding the genetic components affecting these pharmacokinetic processes can help the clinician tailor treatment for a patient.
Slide 8
Chromosomes
Each human has 23 pairs of chromosomes.
Twenty-two are autosomal and look the same in males and females, and one pair is the sex chromosome in which females have two X chromosomes and males have an X and a Y chromosome.
These chromosomes reside in the nucleus of a cell; each chromosome can have hundreds or thousands of genes.
Each chromosome is composed of DNA, which carries the genetic information for the individual.
Twenty-two are autosomal and look the same in males and females, and one pair is the sex chromosome in which females have two X chromosomes and males have an X and a Y chromosome.
These chromosomes reside in the nucleus of a cell; each chromosome can have hundreds or thousands of genes.
Each chromosome is composed of DNA, which carries the genetic information for the individual.
Slide 9
Human Chromosomes
Slide 10
Genes
Composition: Genes only make up about 1% of the total DNA found in humans.
Function: Genes function to produce proteins involved in the millions of biological processes that support the function of the body every day.
Malfunction of genes
Monogenic disease: Single gene malfunctions.
Polygenic disorders: Multiple genes cause disorder; they may have multiple biomarkers.
Function: Genes function to produce proteins involved in the millions of biological processes that support the function of the body every day.
Malfunction of genes
Monogenic disease: Single gene malfunctions.
Polygenic disorders: Multiple genes cause disorder; they may have multiple biomarkers.
Slide 11
Building Blocks of DNA (Nucleotide Bases)
Two purines
Adenine (A)
Guanine (G)
Two pyrimidines
Thymine (T)
Cytosine (C)
Adenine (A)
Guanine (G)
Two pyrimidines
Thymine (T)
Cytosine (C)
Slide 12
Single Nucleotide Polymorphisms
Definition
Single nucleotide polymorphisms (SNPs) are variation in the DNA sequence that differs between members of a species or paired chromosomes in an individual.
Effect on drug activity and metabolism
Many of the genes responsible for drug activity and metabolism (e.g., cytochrome P-450 [CYP]) have different alleles on the same gene, producing different metabolic effects.
Single nucleotide polymorphisms (SNPs) are variation in the DNA sequence that differs between members of a species or paired chromosomes in an individual.
Effect on drug activity and metabolism
Many of the genes responsible for drug activity and metabolism (e.g., cytochrome P-450 [CYP]) have different alleles on the same gene, producing different metabolic effects.
Slide 13
Question #1
Which of the following is INCORRECT regarding the concept of SNPs and differing alleles?
A. SNPs can mutate or malfunction causing diseases.
B. SNPs have different alleles on the same gene producing different metabolic effects.
C. All SNPs in the human body are responsible for turning genes on or off.
D. SNPs can be indicators of future development of diseases.
A. SNPs can mutate or malfunction causing diseases.
B. SNPs have different alleles on the same gene producing different metabolic effects.
C. All SNPs in the human body are responsible for turning genes on or off.
D. SNPs can be indicators of future development of diseases.
Slide 14
Answer to Question #1
All SNPs in the human body are responsible for turning genes on or off.
Rationale: Many of the genes responsible for drug activity and metabolism (e.g., cytochrome P-450 [CYP]) have different alleles on the same gene, producing different metabolic effects. Each person typically inherits two alleles of each gene: one from the mother and one from the father. There can be up to 10 million SNPs in humans, but only those SNPs on coding regions of the gene or the area of the DNA responsible for turning genes on or off have an effect on humans.
Rationale: Many of the genes responsible for drug activity and metabolism (e.g., cytochrome P-450 [CYP]) have different alleles on the same gene, producing different metabolic effects. Each person typically inherits two alleles of each gene: one from the mother and one from the father. There can be up to 10 million SNPs in humans, but only those SNPs on coding regions of the gene or the area of the DNA responsible for turning genes on or off have an effect on humans.
Slide 15
Clinical Applications of Pharmacogenomics
Patients can be given a therapy that inefficiently treats the underlying cause of the disease (lack of therapeutic effect).
Patients can be given a medication that can lead to adverse drug reactions (ADRs) that can be harmful or even fatal due to a difference in their genetic makeup.
Patients can be given a medication that can lead to adverse drug reactions (ADRs) that can be harmful or even fatal due to a difference in their genetic makeup.
Slide 16
Clopidogrel Metabolism and Polymorphism #1
The CYP2C19 gene has nine exons and is situated on chromosome 10.
To date, more than 30 different SNPs have been identified for this gene.
Clopidogrel is given as an inactive prodrug that is rapidly converted to its active metabolite via hepatic bioactivation through CYP2C19 enzymes.
Clopidogrel inhibits adenosine diphosphate-mediated platelet activation and aggregation by irreversibly binding to the platelet purinergic receptor P2RY12.
To date, more than 30 different SNPs have been identified for this gene.
Clopidogrel is given as an inactive prodrug that is rapidly converted to its active metabolite via hepatic bioactivation through CYP2C19 enzymes.
Clopidogrel inhibits adenosine diphosphate-mediated platelet activation and aggregation by irreversibly binding to the platelet purinergic receptor P2RY12.
Slide 17
Clopidogrel Metabolism and Polymorphism #2
About 15% of clopidogrel is modified into an active compound and 85% is hydrolyzed to inactive forms to be excreted.
The metabolism of clopidogrel to its active metabolite is critical to successful treatment, and thus, inherited genetic polymorphisms associated with CYP2C19 have a high impact on the physiological responses to clopidogrel in patients.
Genetic variants of the CYP2C19 gene result in either normal, reduced, or absent enzyme activity or can directly lead to an overactive enzyme.
The metabolism of clopidogrel to its active metabolite is critical to successful treatment, and thus, inherited genetic polymorphisms associated with CYP2C19 have a high impact on the physiological responses to clopidogrel in patients.
Genetic variants of the CYP2C19 gene result in either normal, reduced, or absent enzyme activity or can directly lead to an overactive enzyme.
Slide 18
Types of Mutations on CYP2C19 Gene
CYP2C19*1: wild-type allele (normal enzyme activity).
CYP2C19*2: most common loss-of-function variant allele inherited as an autosomal codominant trait that cosegregates mostly to Asian population.
CYP2C19*3: only detected in less than 10% of the Asian population.
CYP2C19*2: most common loss-of-function variant allele inherited as an autosomal codominant trait that cosegregates mostly to Asian population.
CYP2C19*3: only detected in less than 10% of the Asian population.
Slide 19
Distribution of Mutations Affecting Metabolism of Clopidogrel
Poor metabolizers (PM): 2% to 15% carry loss-of-function mutations (*2/*2,*2/*3,*3/*3) on both alleles resulting in significantly reduced or lack of CYP2C19 activity.
Ultrarapid metabolizers (URM): 5% to 30% have a gain-of-function mutation making CYP2C19 more active (*1/*17,*17/*17).
Extensive metabolizers (EM): 35% to 50% carry a normal CYP2C19 gene (*1/*1) without any mutations.
Intermediate metabolizers (IM): 18% to 45% have only one loss-of-function allele (*1/*2,*1/*3).
Ultrarapid metabolizers (URM): 5% to 30% have a gain-of-function mutation making CYP2C19 more active (*1/*17,*17/*17).
Extensive metabolizers (EM): 35% to 50% carry a normal CYP2C19 gene (*1/*1) without any mutations.
Intermediate metabolizers (IM): 18% to 45% have only one loss-of-function allele (*1/*2,*1/*3).
Slide 20
Question #2
A patient mutation of CYP2C19 is documented as (*1/*17). What is the term for a person with this function mutation?
A. Poor metabolizer
B. Intermediate metabolizer
C. Extensive metabolizer
D. Ultrarapid metabolizer
A. Poor metabolizer
B. Intermediate metabolizer
C. Extensive metabolizer
D. Ultrarapid metabolizer
Slide 21
Answer to Question #2
D. Ultrarapid metabolizer
Rationale: URM have a gain-of-function mutation making CYP2C19 more active (*1/*17,*17/*17). PM carry loss-of-function mutations (*2/*2,*2/*3,*3/*3) on both alleles resulting in significantly reduced or lack of CYP2C19 activity. IM have only one loss-of-function allele (*1/*2,*1/*3). EM: 35% to 50% carry a normal CYP2C19 gene (*1/*1) without any mutations.
Rationale: URM have a gain-of-function mutation making CYP2C19 more active (*1/*17,*17/*17). PM carry loss-of-function mutations (*2/*2,*2/*3,*3/*3) on both alleles resulting in significantly reduced or lack of CYP2C19 activity. IM have only one loss-of-function allele (*1/*2,*1/*3). EM: 35% to 50% carry a normal CYP2C19 gene (*1/*1) without any mutations.
Slide 22
Cloidogrel/Genetic Testing #1
The PMs are those who could benefit the most from genetic testing prior to therapy, as stated on package inserts and as suggested by the Food and Drug Administration.
These PMs are incapable of producing the active metabolite of clopidogrel and would suffer from an unsuccessful treatment of their coronary blockage.
Clinical Pharmacogenetics Implementation Consortium (CPIC) also recommends special attention be given to ultrarapid metabolizers and that alternative therapies be used in poor metabolizers to treat their coronary obstruction.
These PMs are incapable of producing the active metabolite of clopidogrel and would suffer from an unsuccessful treatment of their coronary blockage.
Clinical Pharmacogenetics Implementation Consortium (CPIC) also recommends special attention be given to ultrarapid metabolizers and that alternative therapies be used in poor metabolizers to treat their coronary obstruction.
Slide 23
Cloidogrel/Genetic Testing #2
IM are also challenging to treat with clopidogrel as these patients have a higher number of residual platelets, which could lead to adverse cardiovascular outcomes and therefore might also benefit from other forms of therapy.
Slide 24
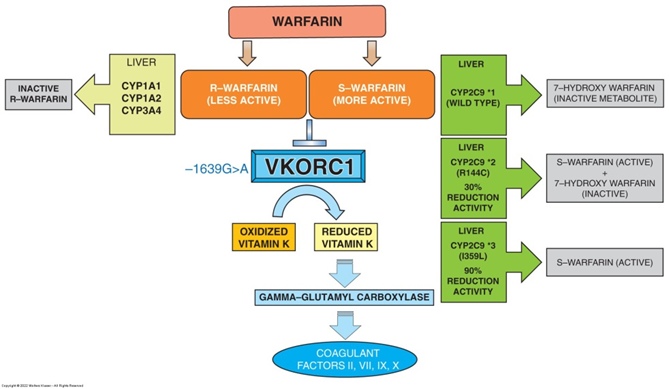
Warfarin Metabolism and Polymorphism #1
Warfarin is a commonly prescribed blood thinner used to prevent atrial fibrillation-induced strokes, as well as permanent damage following the onset of venous thromboembolism or pulmonary embolism.
It exists as a racemic mixture of R-warfarin and S-warfarin.
S-warfarin possesses the most anticoagulant properties through its action as a vitamin K antagonist.
Vitamin K plays a crucial role in the coagulation cascade as its reduced form acts as a cofactor of -glutamyl carboxylase.
It exists as a racemic mixture of R-warfarin and S-warfarin.
S-warfarin possesses the most anticoagulant properties through its action as a vitamin K antagonist.
Vitamin K plays a crucial role in the coagulation cascade as its reduced form acts as a cofactor of -glutamyl carboxylase.
Slide 25
Warfarin Metabolism and Polymorphism #2
This is accomplished by an upstream enzyme called vitamin K epoxide reductase complex, subunit 1 (VKORC1).
VKORC1 is the therapeutic target of warfarin and its inhibition results in decreased amounts of reduced vitamin K preventing coagulation factors from being activated.
Once its therapeutic window is achieved, S-warfarin is rapidly metabolized through the P-450 liver enzyme CYP2C9 to its inactive oxidized form (7-hydroxy warfarin).
VKORC1 is the therapeutic target of warfarin and its inhibition results in decreased amounts of reduced vitamin K preventing coagulation factors from being activated.
Once its therapeutic window is achieved, S-warfarin is rapidly metabolized through the P-450 liver enzyme CYP2C9 to its inactive oxidized form (7-hydroxy warfarin).
Slide 26
Warfarin Metabolism and Polymorphism #2
Slide 27
Metabolism and Polymorphism
The rate at which S-warfarin is metabolized is highly dependent on the enzymatic activity of CYP2C9.
A less efficient metabolism and clearance of warfarin could lead to accumulation of its active form systemically leading to sustained anticoagulation effects.
Deaths have occurred due to excessive bleeding following warfarin treatment. It was later discovered that some patients do not have a fully functional CYP2C9 enzyme due to alleles containing mutations, preventing the proper inactivation of the potent S-warfarin.
A less efficient metabolism and clearance of warfarin could lead to accumulation of its active form systemically leading to sustained anticoagulation effects.
Deaths have occurred due to excessive bleeding following warfarin treatment. It was later discovered that some patients do not have a fully functional CYP2C9 enzyme due to alleles containing mutations, preventing the proper inactivation of the potent S-warfarin.
Slide 28
Genetic Testing to Predict Drug Efficacy and Adverse Response
CYP2C9*1: Individuals possess normal enzyme activity.
CYP2C9*2: Carriers exhibit a 30% decrease in activity.
CYP2C9*3: Patients have as much as 90% decrease in their enzymatic activity.
Patients can carry two normal copies *1/*1 or a normal copy and a polymorphic *1/*2, or could have both copies with polymorphism *2/*3.
The target enzyme VKORC1 has also shown the presence of inactivating mutation -1639G>A.
CYP2C9*2: Carriers exhibit a 30% decrease in activity.
CYP2C9*3: Patients have as much as 90% decrease in their enzymatic activity.
Patients can carry two normal copies *1/*1 or a normal copy and a polymorphic *1/*2, or could have both copies with polymorphism *2/*3.
The target enzyme VKORC1 has also shown the presence of inactivating mutation -1639G>A.
Slide 29
Warfarin/Genetic Testing
For patients receiving warfarin treatment, genetic testing is recommended in order to predict which patients are carrying these mutations who would be at higher risk of bleeding.
A haplotype individual would require less warfarin to inhibit VKORC1 to produce similar anticoagulant effects.
The same is true for patients that carry CYP2C9*2 and *3 where the active form of warfarin does not go through clearance normally leading to immediate excessive bleeding.
A haplotype individual would require less warfarin to inhibit VKORC1 to produce similar anticoagulant effects.
The same is true for patients that carry CYP2C9*2 and *3 where the active form of warfarin does not go through clearance normally leading to immediate excessive bleeding.
Slide 30
Promises, Pitfalls, and Policy Implications #1
NGS: next-generation sequencing
WGS: whole genome sequencing
Exome sequencing
Targeted ribonucleic acid sequencing
Genetic Information Nondiscrimination Act (GINA 2008)
Ethical, legal, and social issues (ELSIs)
WGS: whole genome sequencing
Exome sequencing
Targeted ribonucleic acid sequencing
Genetic Information Nondiscrimination Act (GINA 2008)
Ethical, legal, and social issues (ELSIs)
Slide 31
Promises, Pitfalls, and Policy Implications #2
Another area that is broadly presenting policy challenges involves ethical, legal, and social issues (ELSI) such as patient privacy and protection of data and genetic samples, return of results obtained within the context of research studies to patients, and decisions related to the use of genomic information and insurance coverage.
To date, the implementation of pharmacogenomics into clinical practice has presented with various ELSI and policy challenges.
To date, the implementation of pharmacogenomics into clinical practice has presented with various ELSI and policy challenges.
Slide 32
Summary
Pharmacogenomics and personalized medicine provide both economic opportunities and challenges. The cost of different pharmacogenomic tests continues to decline, and there is increasing evidence for cost-effectiveness of pharmacogenomics, particularly its potential to reduce ADRs.
It is also important to consider how pharmacogenomics might increase costs, including the storage of genetic samples, resources for computational analysis, and interpretation of the findings.
It is also important to consider how pharmacogenomics might increase costs, including the storage of genetic samples, resources for computational analysis, and interpretation of the findings.
Slide 33
The Economics of Pharmacotherapeutics
Slide 34
Learning Objectives
Describe the historical origins and evolution of health insurance and managed care organizations in the United States.
Compare and contrast the common types of prescription drug formularies and corresponding prescription copayment strategies.
Explain the responsibilities of a pharmacy and therapeutics (P&T) committee and describe the factors that P&T committees consider in their decision-making processes.
Discuss how formulary management tools (generic substitution, therapeutic interchange, prior authorization, and step therapy programs) can influence prescribing patterns.
Explain how emerging health technologies (electronic prescribing, electronic medical records, and telehealth) have influenced the delivery of care.
Compare and contrast the common types of prescription drug formularies and corresponding prescription copayment strategies.
Explain the responsibilities of a pharmacy and therapeutics (P&T) committee and describe the factors that P&T committees consider in their decision-making processes.
Discuss how formulary management tools (generic substitution, therapeutic interchange, prior authorization, and step therapy programs) can influence prescribing patterns.
Explain how emerging health technologies (electronic prescribing, electronic medical records, and telehealth) have influenced the delivery of care.
Slide 35
Encouraged the growth of managed care by providing grants and loans to develop Health Maintenance Organizations (HMOs).
Overturned restrictive state laws regulating health providers.
Defined a basic package of services that HMOs were required to offer.
Established procedures by which HMOs could become federally qualified.
Health Maintenance Organization Act of 1973
Defined a basic package of services that HMOs were required to offer.
Established procedures by which HMOs could become federally qualified.
Health Maintenance Organization Act of 1973
Slide 36
Pharmacoeconomic research assesses the "overall value" of medications in the treatment or prevention of the disease(s) they are intended to treat.
Because they evaluate both cost and human data, studies on pharmacoeconomics are important tools for managed care organizations (MCOs) in making drug therapy decisions.
Overview of Pharmaeconomics
Overview of Pharmaeconomics
Slide 37
Cost-benefit analysis
Used to determine the overall cost of a particular intervention or protocol by evaluating all pertinent data and converting the data to a monetary end point.
Cost-minimization analysis
Evaluates the cost of two or more interventions with equivalent components or end points and determines which intervention is least costly.
Types of Analyses #1
Cost-minimization analysis
Evaluates the cost of two or more interventions with equivalent components or end points and determines which intervention is least costly.
Types of Analyses #1
Slide 38
Cost-effectiveness analysis
May help determine the best program or intervention, where the desired outcome is a combination of both a monetary end point and a nonmonetary end point relative to an improvement in health.
Cost-utility analysis
Measures data in terms of quality of life.
These analyses predominantly use quality-adjusted life-years (QALYs) gained as a major outcome.
Types of Analyses #2
Cost-utility analysis
Measures data in terms of quality of life.
These analyses predominantly use quality-adjusted life-years (QALYs) gained as a major outcome.
Types of Analyses #2
Slide 39
Contain a preferred medication list.
Usually organized by therapeutic area and medication class, with the formulary status and reimbursement category listed for each medication.
Encourage the use of medications considered to be safer, more clinically effective, or more cost-effective than other medications within the same therapeutic category.
Formularies and Pharmacy and Therapeutics Committees
Encourage the use of medications considered to be safer, more clinically effective, or more cost-effective than other medications within the same therapeutic category.
Formularies and Pharmacy and Therapeutics Committees
Slide 40
Formularies begin with a basic plan summary and detail key points of reference and then proceed to the list of drugs.
The drugs are most often categorized within their respective therapeutic classes with the therapeutic classes listed in alphabetical order.
Additional information usually detailed for each drug includes the brand/generic status; the drug's relative cost.
A "formulary medication" is a drug that is covered (reimbursed) by the health plan.
Structure of Formularies
Additional information usually detailed for each drug includes the brand/generic status; the drug's relative cost.
A "formulary medication" is a drug that is covered (reimbursed) by the health plan.
Structure of Formularies
Slide 41
Closed: limit clinicians to prescribing from a limited list of preferred agents.
Open: usually do not involve a preferred group of agents; instead, they allow the prescriber to select any covered medication.
Tiered: are open formularies that set different copay tiers for generic, preferred, and nonpreferred medications.
Categories of Formularies
Tiered: are open formularies that set different copay tiers for generic, preferred, and nonpreferred medications.
Categories of Formularies
Slide 42
Which statement describes a characteristic of a tiered formulary?
A. It is based on the patient's ability to pay.
B. It sets different copays for medications.
C. It allows the prescriber to select any covered medication at the same copay.
D. It limits prescribers to a limited list of preferred agents.
Question #1
B. It sets different copays for medications.
C. It allows the prescriber to select any covered medication at the same copay.
D. It limits prescribers to a limited list of preferred agents.
Question #1
Slide 43
B. It sets different copays for medications.
Rationale: Tiered formularies are open formularies that set different copay tiers for generic, preferred, and nonpreferred medications. Closed formularies limit clinicians to prescribing from a limited list of preferred agents. Open formularies usually do not involve a preferred group of agents; instead, they allow the prescriber to select any covered medication.
Answer to Question #1
Answer to Question #1
Slide 44
A portion of the cost of the prescription patients must pay.
Can differ from plan to plan and by geographic region.
Closed and open formularies usually have two copays; one for generics and one for brand drugs.
Tiered formularies have more than two copays, and each tier is related to a copay amount.
Tiered formularies usually have three tiers: generic drugs, preferred brand drugs, and nonpreferred brand drugs.
Copay
Closed and open formularies usually have two copays; one for generics and one for brand drugs.
Tiered formularies have more than two copays, and each tier is related to a copay amount.
Tiered formularies usually have three tiers: generic drugs, preferred brand drugs, and nonpreferred brand drugs.
Copay
Slide 45
Promote the prescribing of safe, efficacious, and cost-effective medications.
Promote the use of less costly and equally efficacious medications (which are most often generic medications).
Financially incentivize the use of preferred brand drugs over nonpreferred brand drugs, or place barriers that prevent nonpreferred branded medications from being covered by the MCO.
Function of Formularies #1
Financially incentivize the use of preferred brand drugs over nonpreferred brand drugs, or place barriers that prevent nonpreferred branded medications from being covered by the MCO.
Function of Formularies #1
Slide 46
Implement policies or programs with their formularies to promote appropriate drug utilization and the use of generic drugs and preferred brand drugs prior to nonpreferred and nonformulary drugs.
Function of Formularies #2
Slide 47
Generic substitution
Therapeutic interchange
Drug-dispensing limitations
Prior authorization and step-therapy programs
Medical necessity
Formulary Policies to Promote Appropriate Drug Utilization
Drug-dispensing limitations
Prior authorization and step-therapy programs
Medical necessity
Formulary Policies to Promote Appropriate Drug Utilization
Slide 48
Structure and function
Composed primarily of physicians and pharmacists and may also include nurses, nurse practitioners, physicians' assistants, and members of the organization's administration.
Formulary management
Develop and revise the formulary, create and implement medication-use policies, and provide education for practitioners.
Pharmacy and Therapeutics (P&T) Committee #1
Formulary management
Develop and revise the formulary, create and implement medication-use policies, and provide education for practitioners.
Pharmacy and Therapeutics (P&T) Committee #1
Slide 49
Development of disease management programs or treatment protocols
These programs and protocols provide useful recommendations for practitioners treating various diseases.
Pharmacy and Therapeutics (P&T) Committee #2
Pharmacy and Therapeutics (P&T) Committee #2
Slide 50
Which statement accurately describes the structure and function of a P&T committee?
A. It is composed of pharmacists and insurance company Chief Financial Officers (CFOs).
B. It licenses pharmacists to practice in hospital settings.
C. It helps to develop and manufacture safe, effective generic medications.
D. It creates and implements medication-use policies.
Question #2
B. It licenses pharmacists to practice in hospital settings.
C. It helps to develop and manufacture safe, effective generic medications.
D. It creates and implements medication-use policies.
Question #2
Slide 51
D. It creates and implements medication-use policies.
Rationale: The main responsibilities of a P&T committee are to develop and revise the formulary, create and implement medication-use policies, and provide education for practitioners. The committee is composed primarily of physicians and pharmacists and may also include nurses, nurse practitioners, physicians' assistants, and members of the organization's administration. The P&T promotes the use of effective generic products but does not develop or manufacture them nor does it license any practitioners.
Answer to Question #2
Answer to Question #2
Slide 52
Prescriber incentives for compliance
Evaluating compliance: percentage formulary compliance
Health care trend reporting
Improving formulary compliance
Pharmacist reimbursement
Patient prescription copayment
Ensuring Formulary and Practice Guideline Compliance
Health care trend reporting
Improving formulary compliance
Pharmacist reimbursement
Patient prescription copayment
Ensuring Formulary and Practice Guideline Compliance
Slide 53
Process whereby a prescriber orders and submits a prescription through an application that electronically transmits the prescription to a pharmacy in real time.
The ability of prescribers to directly transmit prescriptions from the office to the pharmacy eliminates the need for the patient to take it to the pharmacy to be filled.
E-prescribing has been shown to reduce prescribing errors.
Electronic Prescribing and Electronic Health Records
E-prescribing has been shown to reduce prescribing errors.
Electronic Prescribing and Electronic Health Records
Slide 54
Cost of insurance premiums
Affordable Care Act Premises
Ensure accountability of MCOs.
Lower health care costs.
Improve quality of care.
Provide improved consumer choice for health care services.
Current Issues in Managed Care
Ensure accountability of MCOs.
Lower health care costs.
Improve quality of care.
Provide improved consumer choice for health care services.
Current Issues in Managed Care
Slide 55
Despite its critics and shortcomings, managed care is likely to remain the leading manner of financing and delivering health care in the United States.
It is imperative for practitioners and MCOs to understand each other's role in health care.
Although practitioners and MCOs have quite different responsibilities in health care, both groups share a common goal: the delivery of high-quality care to patients.
Summary
Although practitioners and MCOs have quite different responsibilities in health care, both groups share a common goal: the delivery of high-quality care to patients.
Summary
Extracted Images / Illustrations